L7 | CHATS
product update
L7|MASTER®, A Digital No Code / Low Code Authoring Tool to Digitalize Science Across Research, Therapeutics, and Diagnostics
by Robert Zeigler, Ph.D. | posted on August 20, 2024
Today, science is still predominantly planned and documented using paper-based or simple computer-assisted approaches (e.g., Excel), with limited to no standard representations. While we have accepted standards for documents (e.g., Word and PDF) and music files (e.g., MP3), this is not the case when it comes to research, development, diagnostics, and manufacturing processes. Instead, the authoring, collaboration, and distribution of scientific processes and procedures (i.e., protocols) via Word and PDF documents remain the norm, directly hindering digital transformation within the life sciences business.
A Digital Standard to Represent Science
Scientific collaborations across global teams, sites, and organizations, along with the added complexities due to version and revision management, data integrity and regulatory controls, and audit trails, require a digital standard to represent science. This digital standard must be standardized, versioned, qualified, executed, shared, and easily converted to a human-readable format (e.g., PDF). The sharing of a scientific process should be analogous to sharing a song. One does not want to share the sheet music; one rather wants to share and receive the MP3 file that can immediately be played. Establishing the digital standard of science is the only path forward to achieve true digital transformation within the life sciences industry.
L7|MASTER®, a foundational application of the L7 Enterprise Science Platform (L7|ESP®), is a low code/no code authoring tool that enables the creation of any scientific data and process model as a single digital standard, like the MP3 file for music.
THE SOLUTION IS L7|MASTER®
The intuitive user interface provides simplified drag-and-drop features for non-technical users to configure process and data models, thereby reducing the time to define research, development, manufacturing, and diagnostic processes with inherent FAIRification capabilities. Furthermore, the L7|MASTER application provides users with a simplified library of reusable, composable process and data models when creating processes, further achieving data standardization and harmonization across global and interdepartmental organizations.
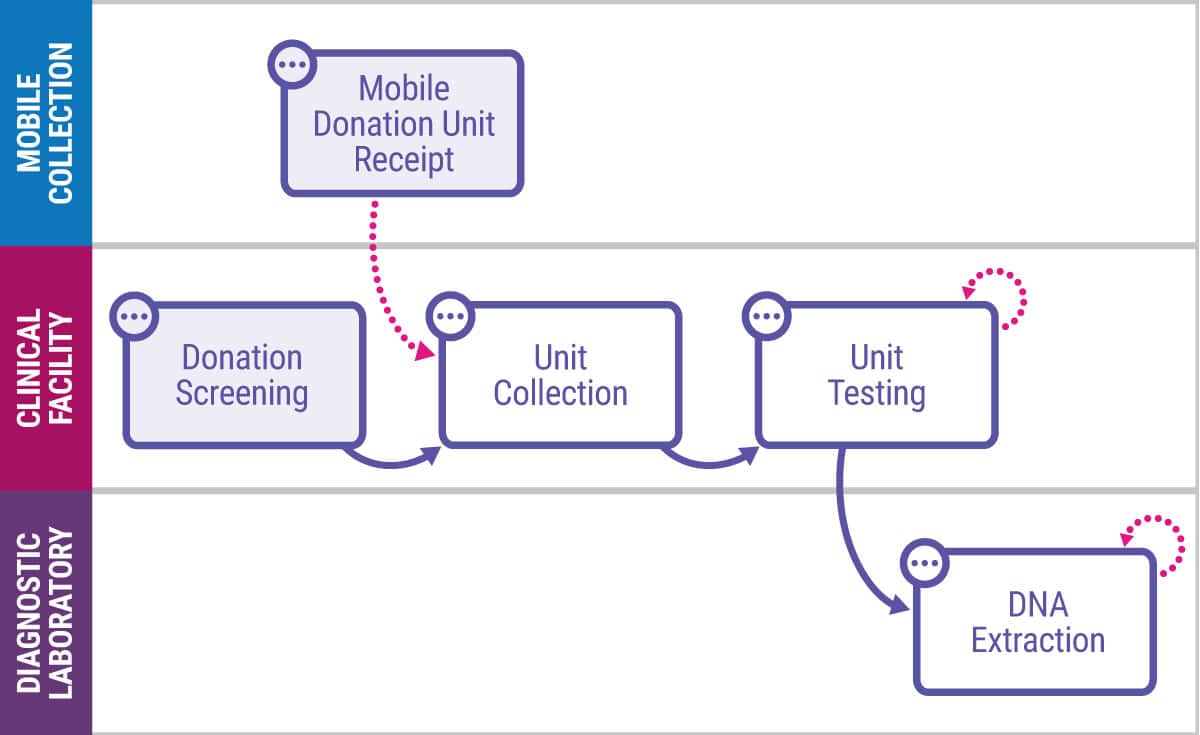
Key Capabilities of the L7|MASTER Authoring Tool
- Creates and manages building blocks for all types of scientific digital processes and process sequences with innate audit-trailed version control.
- Manages all business object types (i.e., master data) and entities (e.g., samples, documents, instruments, and equipment) as they move through workflows regardless of complexity (e.g., feed-forward and feed-back loops).
- Supports integration to ontology management systems and terminology servers to enable better business intelligence, AI/LLM, and predictive modeling.
- Provides a drag-and-drop, low code/no code authoring tool that can be extended via customized HTML code and enables graphical representation of data and process models.
- Offers out-of-the-box data models via an intuitive UI for all traditional aspects of science (e.g., user representation of reagents, freezers, plates, and plate layouts).
- Supports collaborative data and process modeling across scientific teams.
- Supports connectivity to external instruments, equipment, and software systems.
- Generates human-readable PDF documents such as Master Batch Records (MBRs) to enable companies to adopt a digital first framework for modeling science while preserving the ability to distribute procedures as PDF documents.
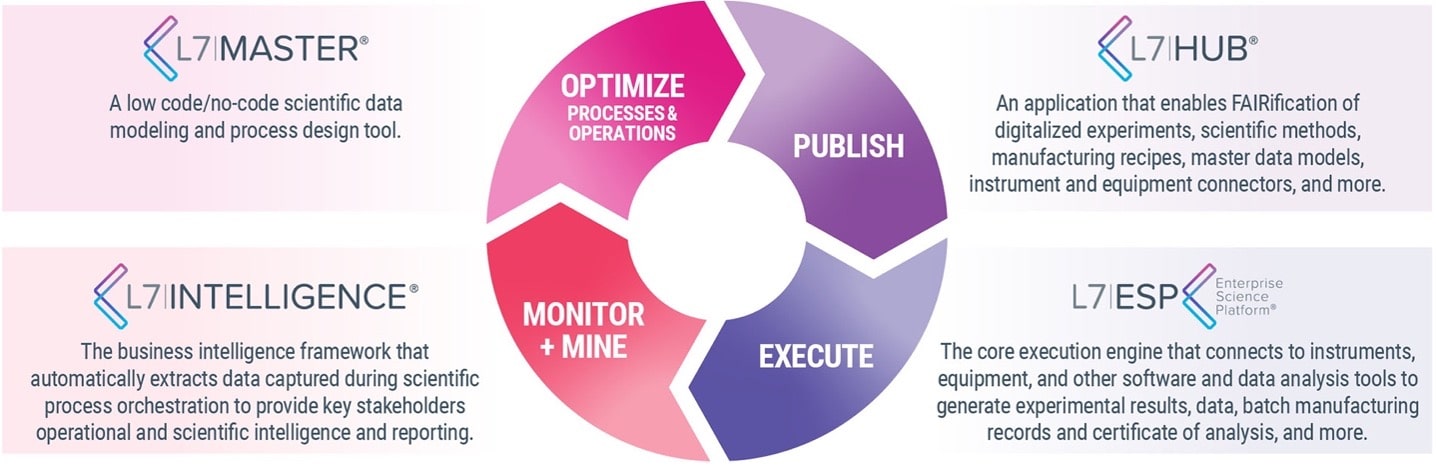
L7|MASTER IS PART OF THE UNIFIED PLATFORM L7|ESP
The L7|MASTER is a key element of the Unified Platform L7|ESP, with its Workflow Orchestration and Data Contextualization, where L7 Notebooks, L7 LIMS, L7 MES, L7 Scheduling, and other scientific data and process management applications reside.
Benefits of the L7|MASTER Authoring Tool
- Accelerate new product introductions (NPIs) at lower cost with the library of ready-to-use standard data and process models to jumpstart the digitalization journey.
- Streamline technology transfers between organizations through DigitalFirst™ collaboration, sharing, and distribution of data within experiments, test methods, master batch records (MBRs), or diagnostic assays.
- Reduce disruption associated with change management, where scientific users can configure and version up scientific processes without dependence on centralized IT teams.
- Reduce the overhead for adoption and training of employees on L7|ESP as digital versions of scientific processes available in L7|MASTER can be directly executed in L7|ESP applications, including L7 LIMS, L7 Notebooks, L7 MES, and L7 Scheduling.
- Reduce TCO (total cost of ownership) as fewer scientists and citizen developers are required to manage a large library of scientific processes.
- Leverage complete audit trails, rapid approvals, version control, and a GMP/GCP/GLP-compliant database to enable risk-based validation.
L7|MASTER is DESIGNED for:
- Research scientists, process engineers, technical writers, Tech-Ops, assay developers, pipeline and process developers, and IT infrastructure and laboratory managers across drug research, development, manufacturing, and clinical diagnostics that use, develop, optimize, and share digitalized scientific data models and protocols.
- Therapeutics research, development, manufacturing, and diagnostics lab managers who need to control the deployment of new digitalized scientific protocols and SOPs.
- CROs and CDMOs who need to streamline technology transfers from one site to another or with their clients and collaborators.
L7|MASTER Got Even Better with Our Latest L7|ESP v3.3 Release
Updates to the L7|MASTER were numerous and included:
- UI/UX enhancements: To strive for optimal user experience, increased UI/UX alignment with design language was considered, of which the result is a builder experience that ensures a consistent editing experience across all the builder L7|MASTER.
- Support of Protocol Pinning and Explicit Protocol Versioning in Workflows: To avoid the introduction of unintentional changes when creating and updating Workflows, Protocols versions can now be given an explicit version name and associated to workflows using that version name, which aids in Change Control compliance and optimizes the maintenance of user Workflows and Workflow Chains.
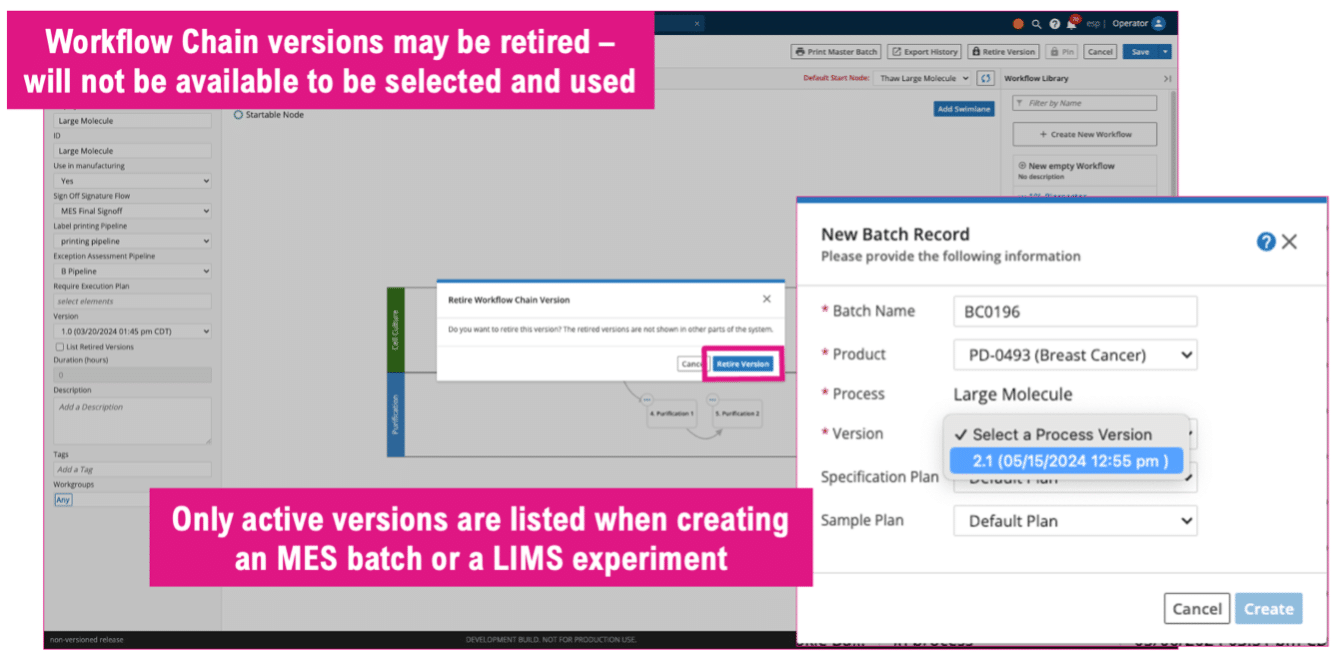
- Improvements to Configuration and Version Control Management: To ensure compliance with the latest set of Standard Operating Procedures (SOPs), workflow version management has been improved to allow users to retire and reactivate WFCs as needed.
- Simplified way to “schedule” entities: Resource availability can be maintained and updated as required, including defining which types of resources can be associated with a scheduled task. This is of particular interest to process/pipeline developers, application platform owners, configuration scientists, lab directors, and project/program managers as it allows them to reserve equipment and other types of resources for use in workflows and monitor available capacity and utilization of those resources.
There are many more exciting L7|ESP 3.3 release updates, which can be found in the L7 Resource Center, and which include:
- Product updates to L7 MES: MES, The Comprehensive Manufacturing Execution System for the Precision Therapeutics Workflow
- Official Release of L7 Scheduling: L7 Scheduling Builds on Embedded Workflows to Maximize Production Capacity and Operational Efficiency
- Product updates to L7 Notebooks: L7 Notebooks Takes ELNs to the Next Level with Embedded Workflows and Dynamic Linking of All Entities
Contact us or visit the L7|ESP landing page to learn more about the L7|ESP platform, with its Workflow Orchestration and Data Contextualization, and how it could help you with your laboratory and business processes.
